Authors
On Monday, another Executive Order (EO) on Drugs was released, this time with an acronym: Most-Favored-Nation (MFN) Prescription Drug Pricing. As expected, the policy aims to address the staggering price differences between prescription drugs in the U.S. and those in our peer nations. While we’re encouraged to see continued momentum around drug pricing transparency, this order is missing some critical components that would give it real teeth. Let’s break it down.
MFN of 2020 vs Today
Although MFN pricing isn't new, this EO introduces some notable differences:
Direct-to-Consumer Mandate
Unlike the 2020 version that focused solely on Medicare beneficiaries and physician-administered drugs, this EO directs the Secretary of Health and Human Services (HHS) to facilitate direct-to-consumer purchasing between patients and manufacturers. This potentially broadens the scope beyond Medicare, but the specifics—such as whether this applies to prescription drugs, physician-administered drugs, or both—remain unclear.
Unclear Pricing Mechanism
The EO instructs HHS to communicate price targets to manufacturers within 30 days. However, it does not outline how MFN pricing will be calculated.
Estimated Savings without Clarity
The EO projects drug price reductions of 30–80%, but it’s vague on methodology and implementation scope. Right now, we only know it targets drugs with “large disparities” in international pricing.
What this means for Pharmaceutical Manufacturers
As the primary stakeholders, pharmaceutical manufacturers are likely to face the steepest challenges:
Revenue Impact
The EO could significantly affect manufacturers’ ability to commercialize current products and fund R&D for pipeline assets. We expect strong industry resistance to delay or derail implementation.
Gross-to-Net Considerations
Companies may need to reevaluate how rebates and list pricing interact. The shift toward a direct-to-consumer model also introduces operational complexity around distribution and compliance.
What this means for Providers and Payers
The ripple effects extend well beyond manufacturers:
Lower Reimbursements for Providers
Aligning U.S. drug prices with international rates could lower reimbursement benchmarks, especially for providers accustomed to the ASP + add-on model. This could erode margins unless providers can offset losses elsewhere in their business.
Impact on Payers
While streamlining the drug supply chain may reduce overall costs, payers could see a decline in rebate-derived revenue. This EO could dramatically shift payer-pharma dynamics.
Guidance vs Plans for Guidance
The intent of MFN is good–we’re all for lower drug costs. However, we’ve been around the regulatory block a few times. Historically, we’ve seen regulations need to have three key elements if they’re to actually make an impact:
- Enforcement dates and clear penalties for non-compliance for any new requirements
- Actual requirements; be it a publicly-available MRF, a report, an estimate tool, etc.
- Technical guidance around requirements
Without these three elements, adherence and real-world impact are fuzzy. For example, the EO states:

This EO does not state new requirements or clear penalties for not lowering costs down to the most-favored targets (or who is included under “other developed countries"), it only says 30 days from now, we’ll see proposed plans for lowering costs. The Trump admin has been hot on transparency (I mean, same) but by our count, we’re racking up a few different proposed regulation deadlines we’ve yet to see come to fruition. It's unclear what will actually happen once guidance is published.
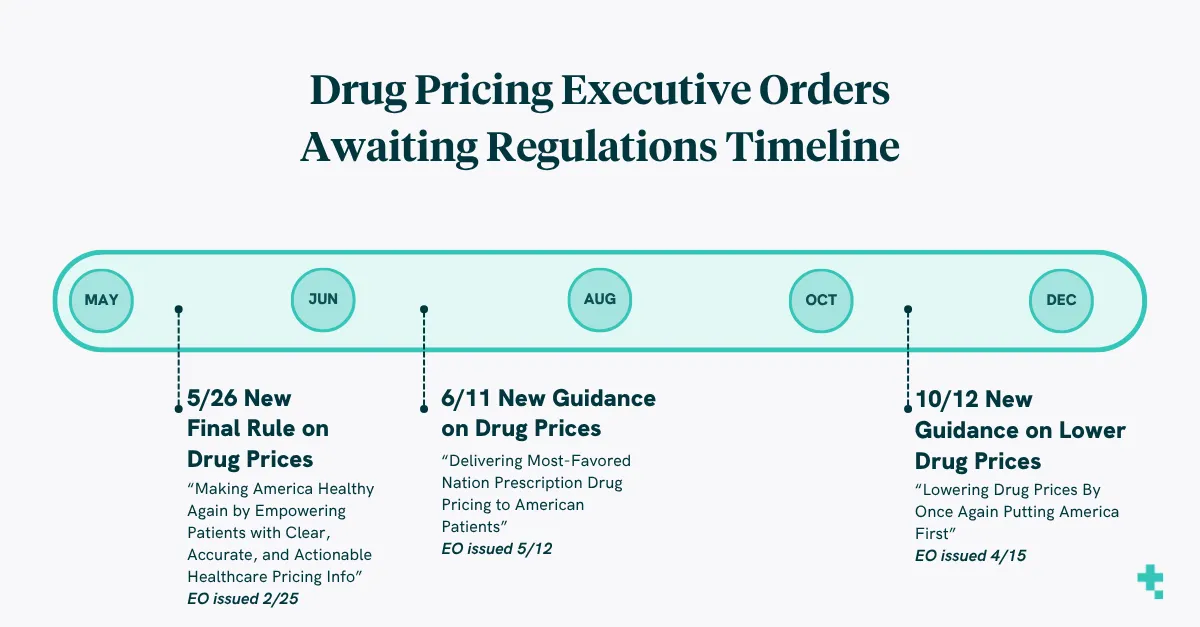
What will drugs cost by October?
In a few short months, the landscape of drug costs may be unrecognizable from today. We are in strong support of any transparency initiatives but we feel that much stronger about initiatives that carry substantive requirements and associated fines. Monitoring over the next few months on what the pushback on this EO is essential, especially on the impact for patient costs and accessibility.
More to come!
See inside the black box
Traceable data, unified workflows, and total transparency
Related resources
Learn, listen, and watch the latest on price transparency.

Introducing the Turquoise Out-of-Network Dashboard
Turquoise launched a free, open license out-of-network pricer built on data designed to eliminate the opacity in QPA calculations, IDR disputes, and broader OON pricing scenarios.


